ZOLGENSMA is a gene therapy designed to treat the genetic root cause of SMA1
Spinal muscular atrophy (SMA) is caused by the deletion or mutation of the survival motor neuron 1 (SMN1) gene. The SMN1 gene produces survival motor neuron (SMN) protein that is critical for normal function of motor neurons. Patients with SMA have an insufficient amount of SMN protein, which leads to permanent loss of motor neurons.2-4
ZOLGENSMA is designed to enable rapid and continuous expression of SMN protein. By replacing the missing SMN protein, ZOLGENSMA can stop the progressive loss of motor neurons.1,5
Elements of ZOLGENSMA enable it to address the cause of SMA1
| Human SMN gene | Continuous promoter | AAV9 vector |
|---|---|---|
| Contains the code for the SMN protein patients need1 | Enables ongoing expression of the SMN gene1,6 | Crosses the blood-brain barrier and enters motor neuron cells1,7 |
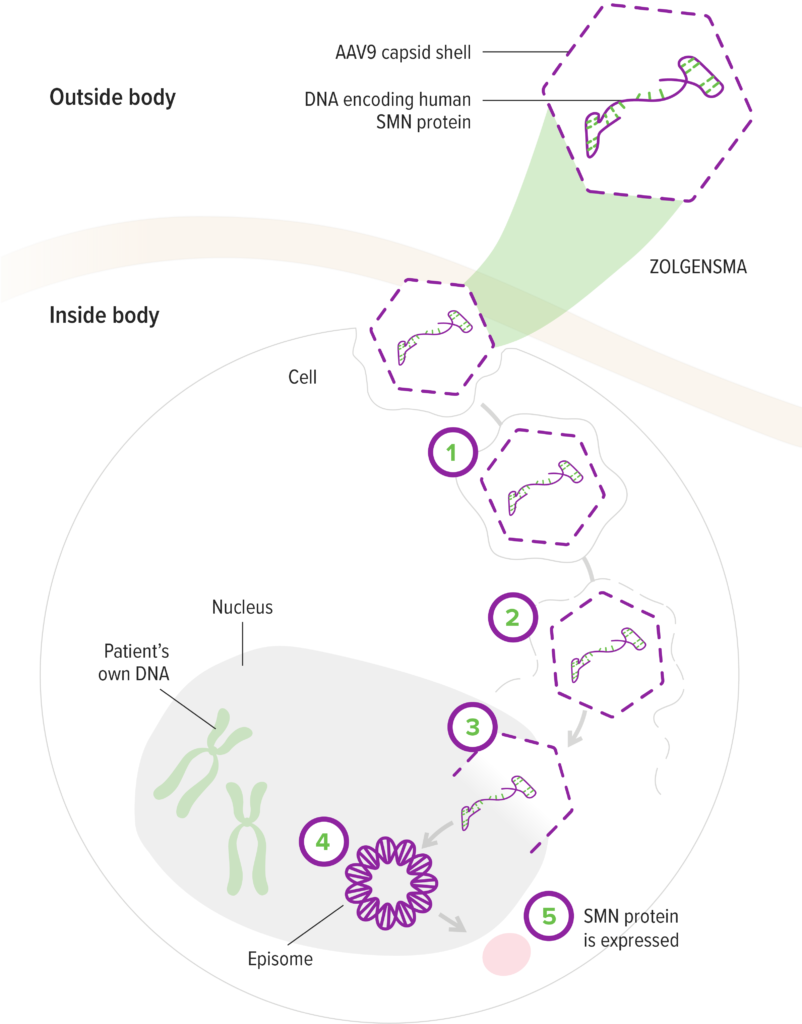
How ZOLGENSMA works
- The AAV9 vector enters motor neuron cells1
- The AAV9 vector delivers the SMN gene to the cell nucleus1,7
- The SMN gene is introduced to target cells as recombinant,
self-complementary DNA1 - The self-complementary ends form a circular episome that can persist in the nucleus of motor neuron cells. These cells are nondividing6,7
- This results in rapid activation and continuous expression of the SMN gene1,5
Watch the process
Transcript
ZOLGENSMA (onasemnogene abeparvovec-xioi) is the first one-time-only gene therapy for the treatment of pediatric patients less than 2 years of age with SMA.
ZOLGENSMA has a Boxed Warning for Serious Liver Injury and Acute Liver Failure. Please see additional Important Safety Information at the end of this video. Please also see the Full Prescribing Information at ZOLGENSMA-hcp.com.
SMA is a progressive and neuromuscular disease resulting from the bi-allelic deletion or mutation in the survival motor neuron 1, or SMN1, gene.
SMN1 is the only gene that consistently produces SMN protein, which is critical for neuronal survival.
SMN1 has a backup gene, SMN2, whose splicing variability results in approximately 10% functional SMN protein.
Without functional SMN1, patients with SMA rely solely on the insufficient levels of protein produced by SMN2.
SMN2 copy number is an important modifier of disease severity in SMA.
In patients with SMA, there is significant variability in SMN2 copy number. Those with the most severe forms of SMA often have 1-2 copies of SMN2.
Without sufficient SMN protein production, patients experience irreversible neuronal loss, resulting in progressive muscle atrophy, which may lead to physical disability, life-threatening medical emergencies, and may result in premature death.
ZOLGENSMA is a gene therapy that stops SMA progression with a one-time-only dose. It targets the genetic root cause of SMA, mutations in the SMN1 gene, by introducing a functional copy of the human SMN gene.
To combat the neurodegenerative nature of the disease, ZOLGENSMA is designed for rapid and continuous SMN protein production, which can stop the progression of disease and preserve motor neurons.
ZOLGENSMA is comprised of a functioning copy of the human SMN gene, which codes for the SMN protein patients need.
ZOLGENSMA has self-complementary ends that form double-stranded loops and has a promoter that activates the human SMN gene and initiates rapid and continuous protein expression.
The genetic elements of ZOLGENSMA are packaged in an adeno-associated virus, serotype 9 or AAV9, vector, which enables delivery of the gene into motor neurons and other, non-CNS tissues.
The AAV9 vector is nonpathogenic, and the viral genes have been removed to decrease immunogenic potential and eliminate potential viral replication. AAV9 vectors are not known to cause disease in humans.
ZOLGENSMA is administered as an intravenous infusion.
Once in the body, the AAV9 vector is able to cross the blood-brain barrier.
Having crossed the blood-brain barrier, the AAV9 vector can efficiently enter motor neuron cells.
Once inside the cell nucleus, the AAV9 vector releases the human SMN gene. The ZOLGENSMA SMN gene is designed to replace the function of the defective SMN1 gene.
The SMN gene is introduced to target cells as recombinant, self-complementary DNA.
The self-complementary ends of ZOLGENSMA are designed to form a circular episome that can persist in the nucleus of motor neuron cells, which are non-dividing.
The self-complementary DNA and continuous promoter allow for ZOLGENSMA to be a one-time treatment by enabling rapid activation and continuous expression of the SMN transgene.
By targeting motor neurons throughout the CNS, ZOLGENSMA stops the widespread neuronal cell death and subsequent muscle degeneration characteristic of SMA, preserving motor neurons and sustaining neuromuscular function.
ZOLGENSMA enables widespread SMN protein expression in the central nervous system (CNS) and in motor neurons6
SMN protein expression was assessed in 2 patients treated with ZOLGENSMA. Expression was detected in viable motor neurons and detected in cells of the brain, as well as the heart, liver, skeletal muscles, and other evaluated tissues.6
Samples were assessed in autopsy. Neither patient death was considered related to ZOLGENSMA treatment. These images were taken of samples from the motor cortex.6
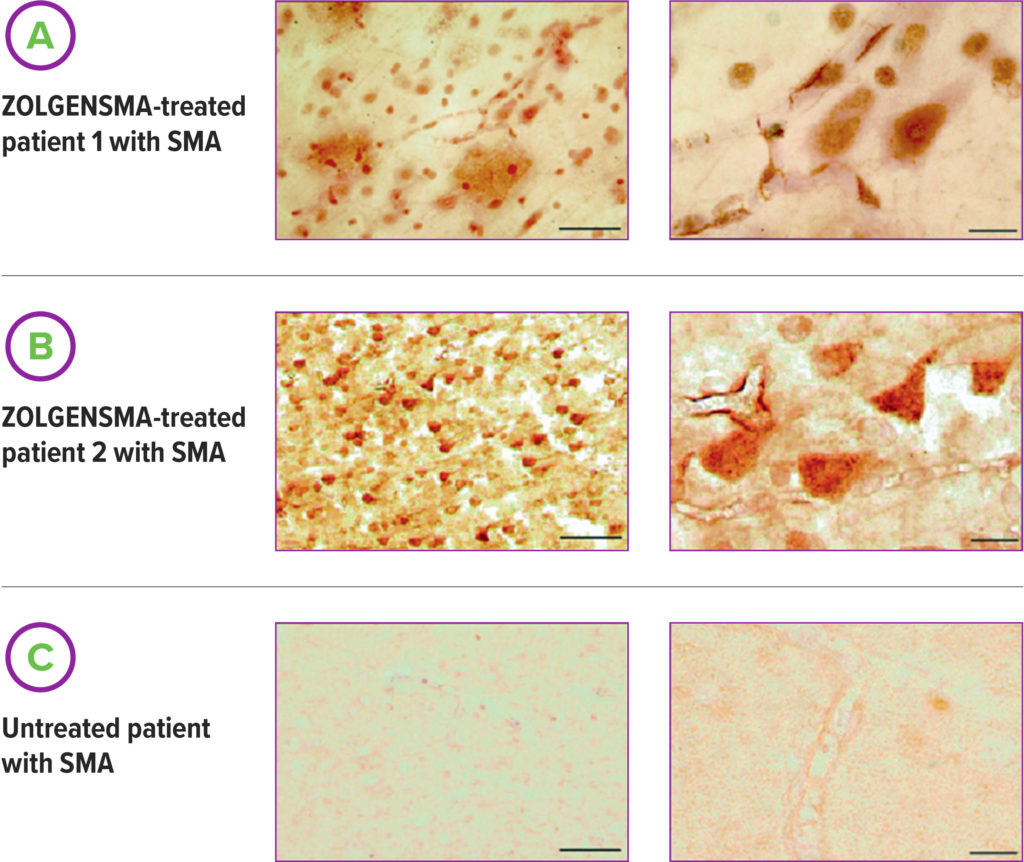
Scale bars (left column)=100 μm; Scale bars (right column)=25 μm.
Reprinted by permission from Springer Nature. Nature Medicine. Biodistribution of onasemnogene abeparvovec DNA, mRNA and SMN protein in human tissue. Thomsen G, Burghes AHM, Hsieh C, et al. 2021.
